
In the United States alone, an estimated 7.9 million adults experience chronic back pain. More details on the recall are available from the FDA.Chronic back pain affects a large portion of the global population, costing billions in direct and indirect medical costs and disrupting the lives of millions of people. The FDA approved Abbott’s Patient Controller smartphone app in 2020. Abbott has distributed a physician communication to reinforce the importance of following the steps regarding MRI scans outlined in the IFU.” “Based on these occurrences, Abbott previously updated the patient controller Instructions for Use (IFU) and patient controller application MRI-mode screen on smartphones reminding the patient to not delete the paired Bluetooth connection between their Implantable Pulse Generator (IPG) and the patient controller. “Abbott has received reports indicating the loss of Bluetooth connectivity between Abbott’s implantable Proclaim and Infinity systems and its paired patient controller after a patient has undergone an MRI,” the company said. For the Clinician Programmer, maintain the paired Bluetooth connection between the IPG and the Clinician Programmer by avoiding deleting the pairing and disabling automatic iOS software upgrades from the iOS settings.Ībbott said in a statement that no devices need to be returned and that physicians can continue to use the Proclaim and Infinity systems. 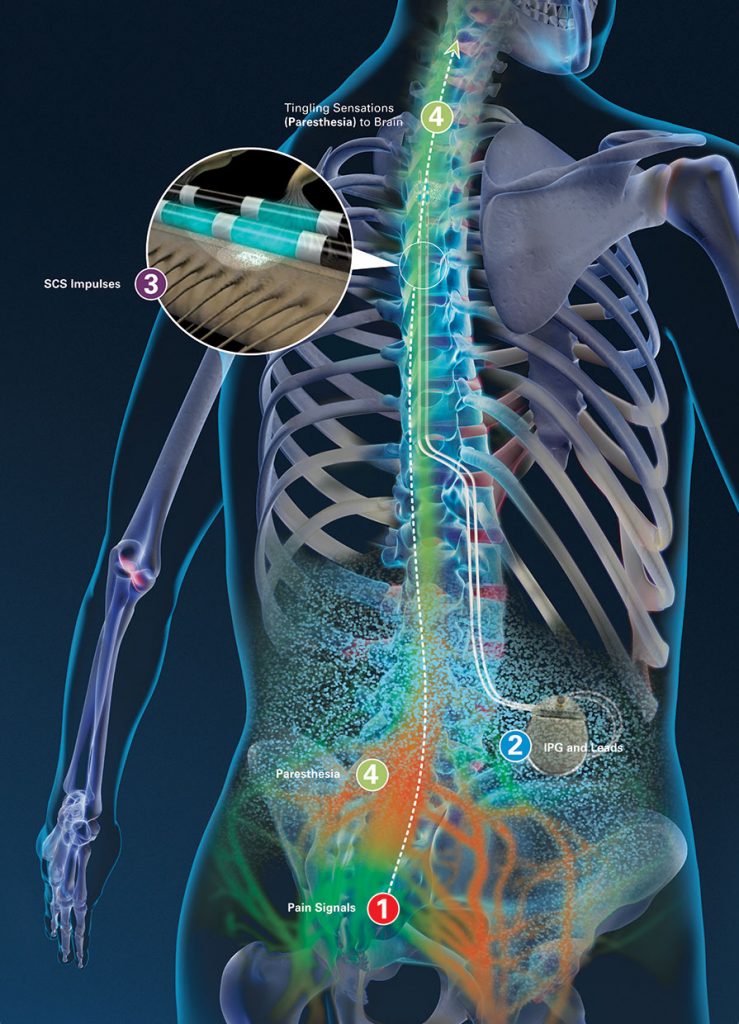
This version of the application provides instructions for the user not to delete the IPG pairing while the system is in MRI mode.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
